Forensic Chemistry Part 1
FORENSIC CHEMISTRY & TOXICOLOGY
CHAPTER 1 – INTRODUCTION
The application of Chemical Principles in the examination of physical evidence falls within the field of Forensic Chemistry. The value of the findings of the chemist in convicting the guilty as well as in clearing the innocent suspect is widely recognized,
Because of that laboratory experts are called upon to render testimony in court.

WHAT ARE THE CAPABILITIES OF FORENSIC CHEMISTRY
The Forensic Chemistry Branch is tasked to examine the chemical nature and composition of the various specimens, such as blood and other body fluids, gunpowder, explosives, poison, dangerous drugs and wines, soil analysis, metallurgy, ink and paper of documents, and many more.
This is how blood looks like under the high-powered microscope.

Gunpowder and explosives…

Poison and drugs…

and many more…
EXAMINATION AND ANALYSIS
Examination of materials submitted may be qualitative (determining what constituents are present) or quantitative (determining how much of the constituents are present).

With the NEW DEVELOPMENT in the field of instrumental analysis, chemists are no longer limited to the so-called “wet” methods, which require much time and effort. For evidence submitted in very small amounts, high precision instruments such as the ultra-violet and infrared spectrophotometer, thin layer chromatography, and the gas chromatograph can be utilized.

To summarize, FORENSIC CHEMISTRY AND TOXICOLOGY deals with the application of chemistry in the identification of physical evidence.

It includes collection, preservation, examination, and study of the following:
[1] blood;
[2] semen and other bodily fluids;
[3] gunpowder and explosives;
[4] hair and textiles fibers;
[5] chemical aspects of document examination;
[6] problems of glass fragments and fractures;
[7] moulage;
[8] metallurgy;
[9] petrography as applied to criminal investigations;
[10] chemical analysis of arson evidence

[11] drugs; and
[12] study of poison and the following:
(a) nature of poison
(b) physiological action,
(c) chemical and physical properties;
(d) dosage, treatment, and detection of poison.
THE FOUR STAGES IN THE PRACTICE OF FORENSIC SCIENCE (CHEMISTRY)
The collection, preservation, and forensic analysis of the evidence with most crimes are often critical in determining a person’s guilt or a person’s innocence. The forensic exam is beneficial to law enforcement and the chemical and biological setting to provide a comprehensive forensic examination with the collection of evidence knowing the victim’s identity and the available evidence at the crime scene.

Evidence must be identified, collected, packaged, secured, and maintained correctly, then released to Law Enforcement following a strict chain of custody rules so that it can be analyzed appropriately and used later in legal proceedings. When collecting evidence, the examiner must wear gloves at all times and change them often utilizing the clean technique.

The following are the STAGES IN THE PRACTICE OF FORENSIC INVESTIGATION.
(1) Collection or Reception of Specimen
Guiding principles are as follows:

(a) Sufficiency of samples
(b) Standard for comparison
(c) Maintenance if individuality
(d) Labeling and sealing
(2) The Actual Examination of Specimen

(3) Communication of the Results of the Examination

(4) Court Appearance

SIX GOLDEN RULES IN THE PRACTICE OF FORENSIC CHEMISTRY
[1] Go Slowly

[2] Be thorough

[3] Take notes

[4] Consult others

[5] Use imagination

[6] Avoid complicated theories

CHAPTER I
POISONING

INTRODUCTION
For hundreds if not thousands of years, the poison of one sort or another has been the weapon of choice among dispassionate murderers. Avoiding the drama and mess that may often be generated by an overly violent death, a carefully chosen poison could rely upon to work slowly, stealthily and ultimately surely.

With the advantage of a little background knowledge and access to the right materials, a murderer might even tailor his weapon to confuse the victim and any subsequent inquiry by using it to produce symptoms resembling those of natural killers, such as heart disease or pneumonia.

But modern scientific methods make it increasingly difficult for the poison to remain undetected. Long ago, in 1814, Mathieu Joseph Orfila earned the title of the “father of Toxicology” by publishing the book “Traire des Poisons,” which classified the common poisons favored by criminals. In 1836, another pioneer, Alfred Swaine Taylor, published a monumental textbook called Elements of Medical Jurisprudence that became a classic of forensic medicine.
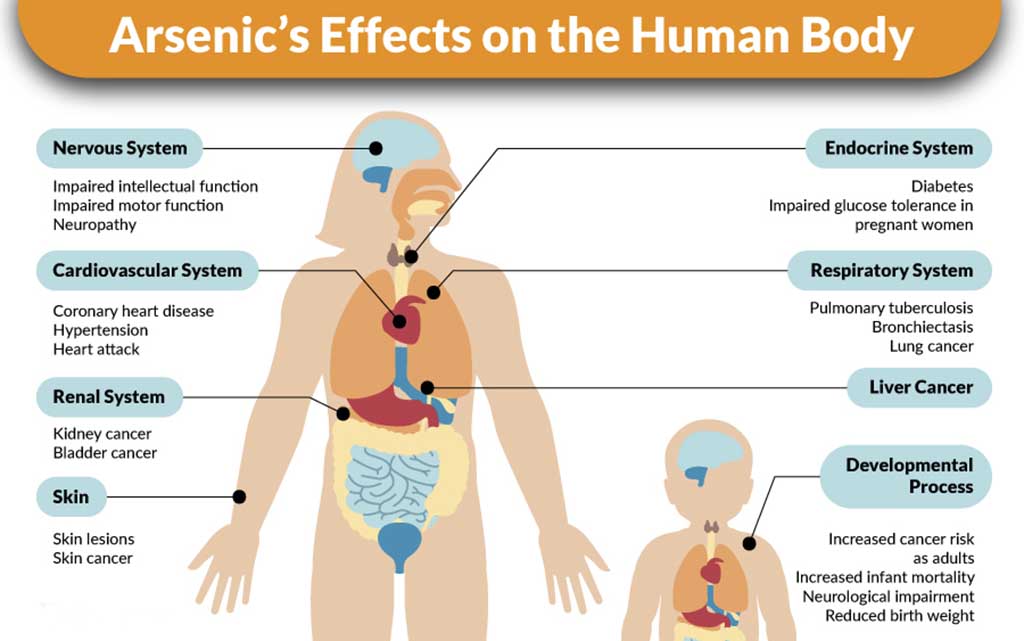
In October of that year, English chemist James Marsh, working at the Royal Arsenal at Woolwich, South London, developed an accurate technique for revealing traces of arsenic. Criminals favored this poison because arsenic already exists in small traces in the healthy human body. A victim of arsenic poisoning, however, has traces of the chemicals in almost every part of the body as opposed to concentrations in particular organs such as the stomach or liver. These remain in the bones and hair even after death, whereas the body’s chemistry may break down other poisons.

Marsh’s test could reveal a trace as small as one-fiftieth of a milligram in a sample taken from the body of a victim of a suspicious death, and the principles of his techniques are still used today.
TOXICOLOGY, POISONING, AND ITS CLASSIFICATION

TOXICOLOGY (TOXIC – poison; OLOGY – science) – is that branch of science that treats poison, its origin, physical and chemical properties, physiological action, treatment of their noxious effects, and detection method.
POISON – is a substance that, when introduced into the body and absorbed through the bloodstream and acting chemically, can produce noxious effects or destroy life.
CLASSIFICATIONS OF POISONS – There are several classifications of poisons based on different standards:
[1] According to Kingdom:
{a} Animal – example is cantharides
{b} Vegetables – an example is a strychnine
{c} Mineral – an example is a hydrochloric acid

[2] According to Chemical Properties
{a} Inorganic Poisons
{1} Volatile and non-volatile; ex. Bromine, chlorine, iodine
{2} Mineral acids – ex. Hydrochloric acid
{3} Mineral alkalies – ex. Sodium hydroxide

{b} Organic Poison
{1} Volatile – ex. Alcohol, chloroform
{2} Alkaloid – ex. Strychnine, brucine
{c} Animal poisons – ex. Snake venom
{d} Bacterial – ptomains
{e} Organic acids – ex. Salicylic acid
{f} Glucosides – ex. Digitalis
[3] According to Physiological Action
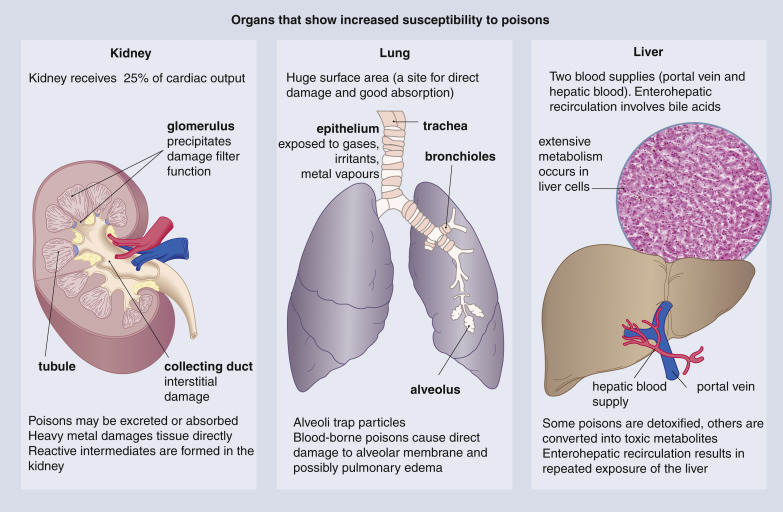
(a) Corrosives
A highly irritant poison that causes local destruction of tissues and is characterized by nausea, vomiting, great local distress. Example Strong Acids and Alkalies.
(b) Irritants
One which produces irritation or inflammation of the mucous membranes and is characterized by vomiting, pain in the abdomen, and purging. Ex. Arsenic, croton oil, cantharides.
(c) Neurotics
One which acts chiefly on the nervous system producing delirium, convulsions, and coma, with disordered circulation and respiration as the outstanding symptoms. Ex. Alcohol, opium, strychnine, carbon monoxide.
(d) Narcotics
Poisons that produce stupor, complete insensibility, or loss of feeling. Ex. Opium, Demerol, cocaine, and cocaine substitutes.
(e) Tetanics
Substances that act directly upon the spinal cord producing such spasmodic and continuous contraction of muscles as a result of stiffness or immobility of the parts to which they are attached. Ex. Nux vomica, strychnine, brucine.
(f) Depressant or sedatives
Agents which retard or depress the physiological action of an organ. Ex. Antipyrine, nicotine, and cocaine.
(g) Asthenic or Exhaustive
Agents which produce exhaustion; marked loss of vital or muscular power. Ex. Aconite, digitalis, hydrocyanic acid.
[4] According to Pharmacological Action:
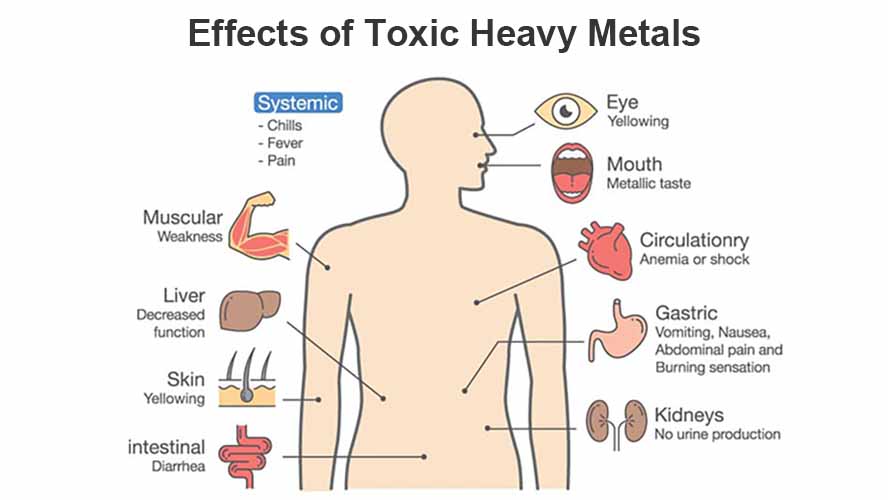
(a) Substances characterized by local action. Examples: volatile oils, skin irritants, and purgatives.
(b) Substances are characterized chiefly by their action after absorption. Examples are alkaloids and glucosides.
(c) Heavy metals and metalloids. Examples: phosphorous and arsenic.
[5] According to the Method of Isolation
(a) Volatile poisons and those isolated by distillation with or without current or stream.
Example: alcohol, phenol-chloroform.
(b) Non-volatile poisons – those are isolated by extraction with organic solvents.
Examples: alkaloids, synthetics, and organic acids.
(c) Metallic poisons – for example, arsenic, mercury
(d) Substances for which special methods of isolation are required.
For example, acids and alkalies and salt of alkali metals are extracted with water; oxalic acids and those requiring dialysis.
TYPE OF POISONING
[A] From the legal point of view
1. Accidental
2. Suicidal
3. Homicidal
4. Undetermined
[B] From the medical point of view
1. Acute poisoning
2. Sub-acute poisoning
3. Chronic poisoning
ACTIONS OF POISONS
[1] Local – the changes or disturbance produced on that part with which the poison comes in contact. For example, erosion is produced by corrosive poisons.
[2] Remote – the changes or disturbance produced in distinct parts away from the site of the application. For example, dilation of pupils when belladonna is taken orally.
[3] Combined – the effect of the poison is not only localized at the site but also affects remote organs, for example, phenol causes corrosion of the gastrointestinal tract (local) and causes convulsions (action on the cord)
CONDITIONS MODIFYING THE ACTION OF THE POISONS
Those attributed to the individual:
1. Age and sex
2. Health and vigor
3. Habit
4. Idiosyncrasy
5. Disease
6. Food
7. Sleep
8. Exhaustion
Those which are attributed to the poison:
[1] Physical state or form of the poison
[2] Direction
[3] Dose – the quantity of poisons to be administered at one time. The branch of science which treats the form and quantity to be administered within a certain period is called POSOLOGY.

[4] Solubility of poisons
[5] Mode of administration
[6] Chemical combination
[7] Mechanical combination
[8] Condition of the surface where the poison was applied.
ENTRANCE OF POISONS (Methods of Administration)
[1] Mouth and are absorbed into the circulation after passing through the stomach and intestinal wall.
[2] Nose and enter the blood from the upper respiratory passages or lungs.
[3] Eyes
[4] Rectum, vagina, urethra, bladder & urether by injection.
[5] Hypodermic injection
[6] Intravenous injection
ELIMINATION OF POISONS
1. Emesis
2. Respiration
3. Feces
4. Urine
5. Milk
6. Sweat
7. Saliva
8. Tears
DIAGNOSIS OF POISONING
(1) Systems of poisoning come suddenly upon a person who previously has been in good health, while the disease is usually preceded by a number of hours, days, or even weeks of local or general indisposition.
(2) In cases of poisoning, the symptoms commonly make their appearance after taking food, drinks, or medicine.
(3) If several persons take the same food and drinks, they should all show similar
(4) Diseases are generally slower in their progress and are preceded by circumstances as exposure, recognized symptoms, and general or local indisposition of longer duration.
GENERAL TREATMENT OF POISONING
[1] Removal of the poison from the stomach or the body.
(a) If applied or instilled – washing
(b) If injected – ligatures and bleeding
(c) If taken orally
[2] Administration of antidotes – an antidote is any agent which neutralizes a poison or otherwise counteracts or opposes or its effects.
(A) Kinds of antidotes:
i. Mechanical antidotes
(a) Stomach tube or pump
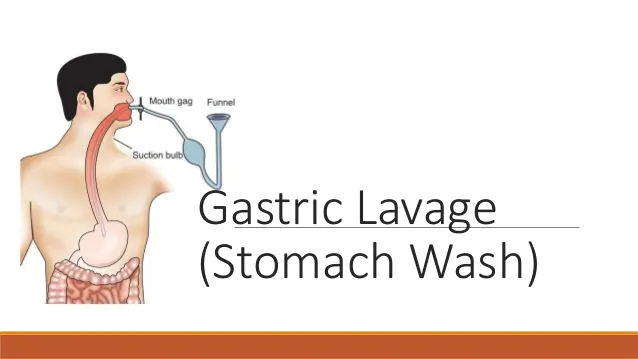
(b) Emetics
1. Local emetics
2. Systems or general emetics
3. Cathartics
4. Demulcents
5. Precipitants
ii. Chemical antidotes
iii. Physiological antidotes
(a) Elimination of Poisons by Excretion
(b) Elimination of Other Symptomatic Treatment
TREATMENT FOR METALLIC POISONS
Fresenius and Von Babo Methods or ashing or by dry fusion.
Kinds of metallic poisons are:
[1]Arsenic – Prelim test – by Reinsch Test/Confirmatory – by Marsh and Gutzeith test.
[2] Lead
[3] Mercury
[4] Thallium
[5] Antimony
[6] Bismuth
End of Chapter 1
CHAPTER 2

The main function of blood is to transport oxygen, nutrients, and wastes throughout the body. Blood is approximately eight (8%) percent of the body’s weight. It consists of the liquid portion called plasma (about fifty-five to sixty-five percent [55-65%] of the total blood volume) and the formed elements, which are the red blood cells (erythrocytes), the white blood cells (leukocytes), and the platelets (thrombocytes).

The hemoglobin found in the red blood cells is responsible for transporting oxygen to tissues and carbon dioxide to the lungs. The serum is the clear liquid that separates when the blood is allowed to clot. If blood is allowed to stand for quite some time in which and oxalate is added the formed elements settled down and a straw-colored liquid called plasma is formed at the top.

BLOOD AS IMPORTANT EVIDENCE IN INVESTIGATION
An adult has some ten pints of blood circulating around the body, driven under pressure from the heart’s pumping action. This presents a potential attacker with something of a problem. Any cutting or piercing of major blood vessels can produce a deluge and result in significant traces of blood being left at the crime scene. Such traces can indicate how an attack was made and help identify all manners of objects associated with it. Even the attacker’s own blood may lead to his or her identification in a violent struggle and can tie him or her to the crime as securely as a photograph, scientific analysis, or eyewitness identification.
For these reasons, criminals can often go to great lengths to remove blood stains from a crime scene. They may satisfy themselves that all traces have been eliminated, but once the crime has been discovered the police search usually extends to areas beyond the perpetrator’s control. Forensic experts check beneath washbasins and inside drawers, between floorboards or in drains, and waste pipes leading from baths or sinks that may have been used for washing or for cleaning the hands and fatal weapons.

At one time, blood found on the crime scene was significant to forensic scientists simply by its presence. Because blood tends to dry quickly on exposure to air and to remain clearly visible unless cleaned away, bloodstains and splashes can often tell their own story.
Drops of blood, therefore, are very much useful because their characteristics may explain some of the nature of how a crime happens. Stains made by blood spurting under pressure from major blood vessels show where a serious or even fatal blow was delivered.
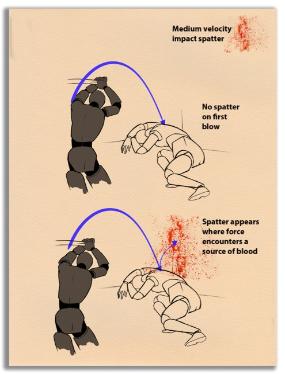
The height reached by the spurting blood, if it is splashed on a wall or partition, for example, can show whether the victim was standing, sitting, kneeling, or lying down when the fatal blow was initiated. The quantity of blood spilled, when compared with injuries to the victim’s body, can be correlated with particular wounds. Pools of blood can also indicate where the victims died, even if the body was later removed.
IMPORTANCE OF THE STUDY OF BLOOD
[1] As circumstantial or corroborative evidence against or in favor of the perpetrator of the crime;
[2] For disputed parentage;
[3] Determination of the causes of death, and the length of time the victim survived the attack;
[4] Determination of the direction of escape of the victim;
[5] Determination of the origin of the flow of blood;
[6] Determination of the approximate time the crime was committed.
ACTUAL EXAMINATION OF BLOOD
Determining if the Specimen is Blood
The following examination are conducted: A preliminary examination and a confirmatory examination to determine that it is actually blood; and an analysis to determine the origin and grouping test to determine the blood group.
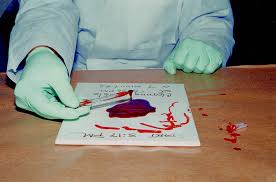
Preliminary Examination or Screening Test
To rapidly screen the evidence for the presence of blood, the BENZIDINE TEST is done. Although there are numerous chemical tests available, the benzidine test is done because it is very sensitive and can be easily performed even by investigators in the field. In the case that the benzidine test cannot be conducted, the investigator should try the PHENOLPHTHALEIN TEST.
Confirmatory Test whether a Stain is a Blood
There are two tests that can be used to further established the presence of blood: the HEMIN (Teichman) test and the HAEMOCHROMOGEN (Takayama) Test. Both tests are designed to show the presence of hemoglobin in a suspected bloodstain by the appearance of specific crystals observed under the microscope.
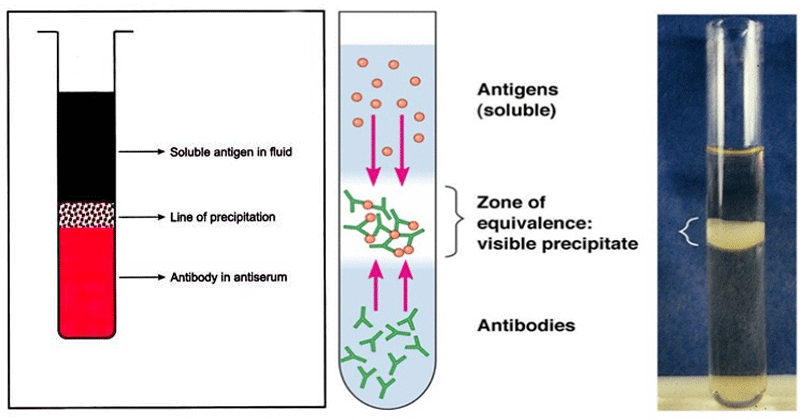
After confirmation that the stain is blood, the investigator should determine whether the blood is of human or animal. This may be done through PRECIPITIN TEST. The precipitin reaction distinguishes between the blood of a human being and that of an animal. The test is based on the fact that when an animal’s blood is injected with the protein of an animal of a different species, an antibody is developed in its serum, which causes the latter to react specifically with the protein of any other species. A band (zone of equivalence) or ring that forms between the blood specimen only indicates that blood does not come from the same origin.
Factors Affecting the Precipitin Reaction:
[1] Age of stain and putrefaction – can cause changes in blood, though often not severe;
[2] Exposure to air sunlight and humidity – hastens oxidative changes making a conclusive identification difficult;
[3] High Temperature – cause weaker and extended precipitation;
[4] Contamination of chemicals such as soap or detergent may interfere or cause false-positive reactions.
Grouping Test of Blood
To determine to which major human blood group confirmed human blood belongs, grouping tests are conducted. These are based on the ability of the blood serum of one person to clump or bring together the red blood cells of certain individuals. If a suspension of human red blood cells is mixed with its own serum, or the serum of a similar group, the cells remain even.

But if they are mixed with the serum of another group, the cells clumps or agglutinate. This process is called AGGLUTINATION, which was first observed by Landsteiner in 1900. This was explained on the basis that the red blood cells (RBC) contain an antigen or agglutinogen and that the serum contains antibodies or agglutinins.
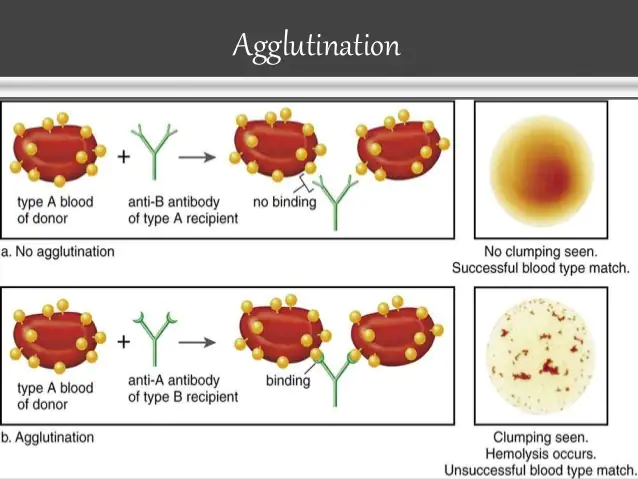
Two agglutinogens and two agglutinins were found, which are designated for convenience, A, B, anti-A, and anti-B respectively. This became the basis for the ABO System. Later researches discovered other agglutinogens and agglutinins and other systems of classifying types of blood were made.
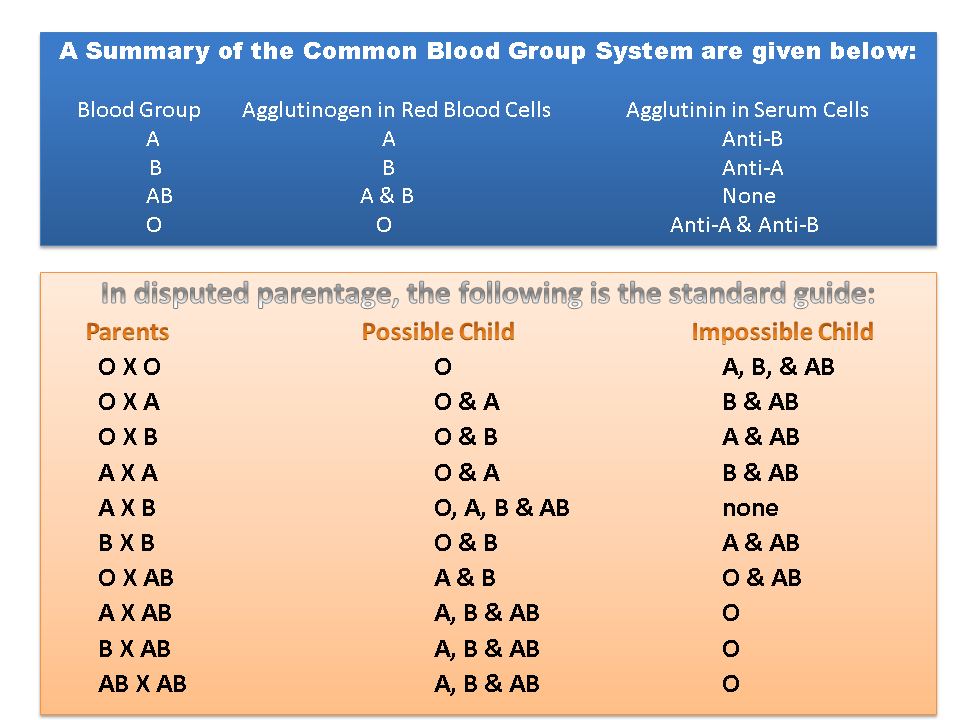
Under the foregoing, if the blood groups of husband and wife are both O. then those of the offspring are all O and is impossible to have children with A, B & AB blood group, And so on…
OTHER BODILY FLUID
It has been found that some individuals secrete in their fluid – such as semen, saliva, urine, and vaginal fluids and substances corresponding to their blood groups. Therefore, it is possible to determine the ABO blood group of suspected stains of body fluids from the secretors.
Blood also extracted from individuals suspected to be intoxicated are examined to determine whether they are under the influence of alcohol or not, at the time of the extraction. The percentage of alcohol obtained from the blood is examined and interpreted by the medico-legal officer to determine the percentage of alcohol level in the blood.
THE PERISHABLE NATURE OF BLOOD
Blood is a type of evidence, which undergoes a rapid change in its character with the passage of time, as the process of clotting and drying commences almost immediately on exposure to air. Furthermore, blood offers little resistance to decomposition, especially when exposed to certain conditions and influences, which if prolonged, will cause the specimen to lose its identity.
It is therefore extremely important that blood samples be sent to the laboratory as soon as possible after their discovery or scientifically preserve. Prior to the general search of the crime scene, the floor should be examined carefully in order to minimize the danger of loss or destruction caused by being walked (contamination is the most common) by persons otherwise engaged in the investigation. There are several approaches today that could also enhance, identify and locate the presence of blood despite being washed, see our example below.
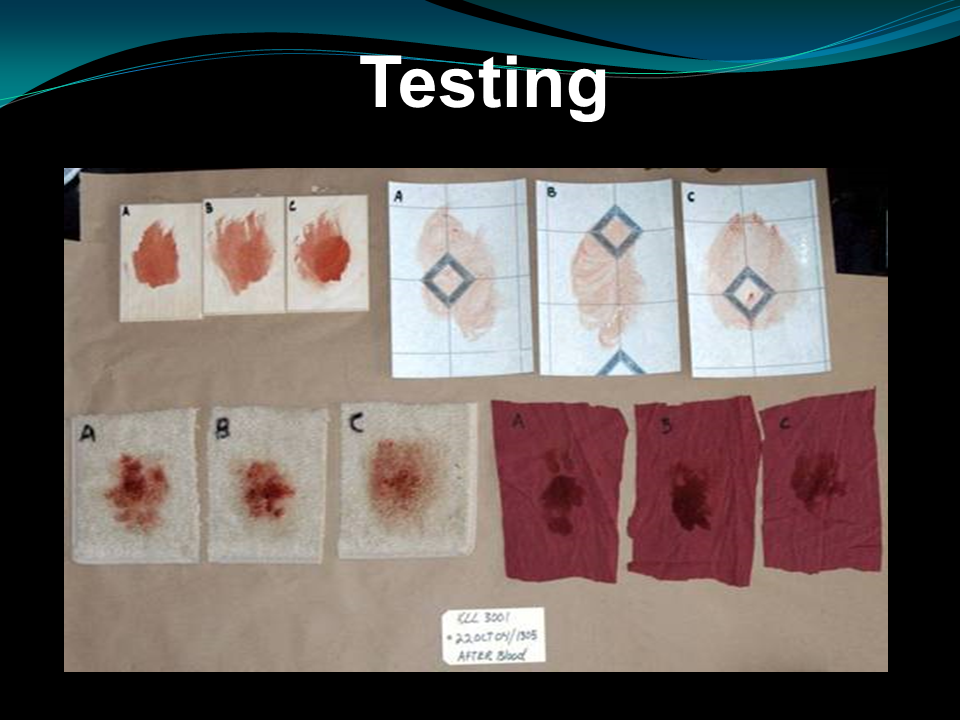
Above specimens are treated differently by washing rubbing and painted over, then look at the result, despite the efforts of defacing it from its surface:
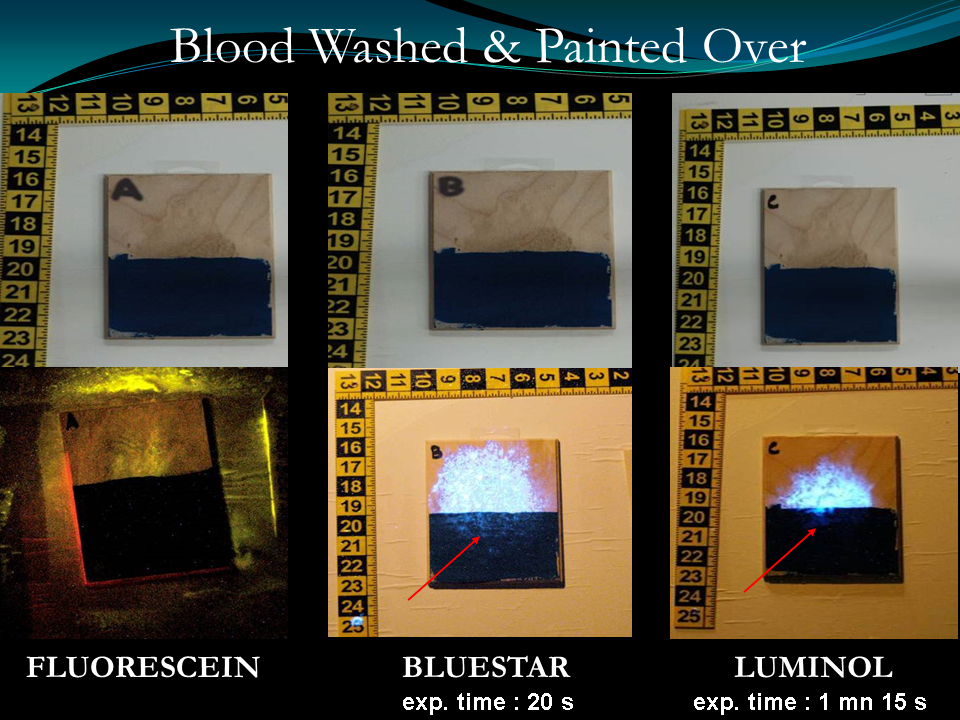




All example above is a proof that blood can be detected very well on every surface even efforts are being made to erase it. Technically and scientifically only a well-learned and trained investigator can do this.
End of Chapter 2
CHAPTER 3
THE SEMEN AND ITS COMPOSITION
Semen refers to the fluid produced by the male sex organ. It is usually white to yellowish in color, consisting of two parts: the seminal plasma or fluid, and the spermatozoa or sperm cells. There are usually 70,000,000 to 150,000,000 sperm cells per milliliter of semen (And a healthy young man may produce 400-500 million spermatozoa per ejaculation. And normally, in a single ejaculation 1.5 to 3.5 cc are being produced.
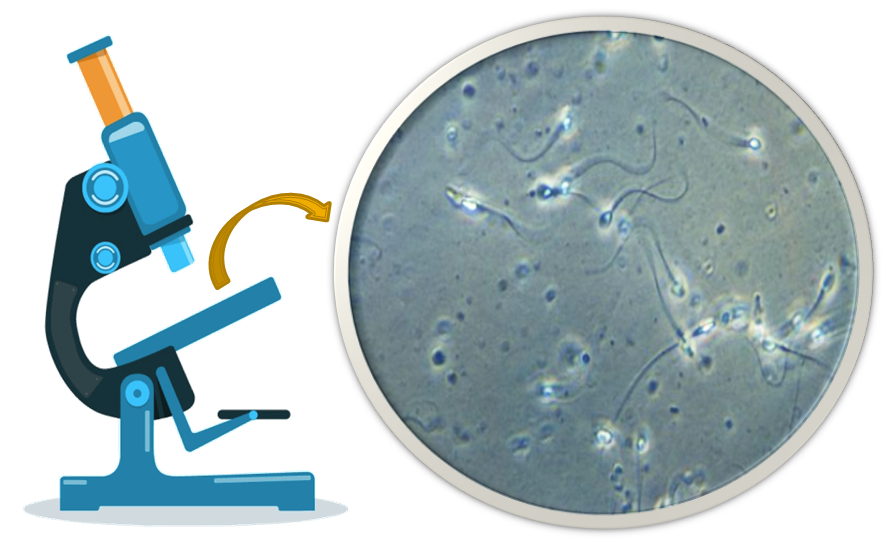
The sperm cells, as seen under a microscope consist of a head, a neck, and a tail approximately ten times as long as the head. During deterioration, the bacteria attack first the tail, making identification difficult. The seminal fluid contains certain substances called flavins, which help give a yellowish color to semen and cause it to flourish under ultra-violet lights.
THE SEMINAL FLUID
In cases of rape and other sexual crimes, the seminal fluid left by the attacker on the victim’s body or on clothing or furniture presents another powerful source of evidence. Since the sperm in the fluid remain alive for only a relatively short period, the condition of the sample can give reasonably accurate indications of the time of the attack. In the case of secretors, such as sample contains information of blood group and the presence or absence of other enzymes and proteins that can help to concentrate the search for potential suspects nor rather perpetrators.
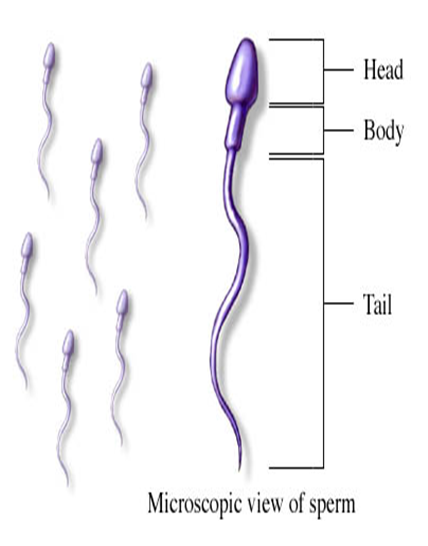
Samples of seminal stains or fluid are isolated using tests similar to those used to reveal the presence of bloodstains. These tests are particularly useful where attempts have been made by the criminal to remove or wash away incriminating traces. The preceding discussion is intended to explain what are tests are.
EXAMINATION FOR SEMEN
(1) WET SPECIMEN
Normally, suspected semen may be found undried having the alkaline odor characteristics for seminal fluid. In this condition, an examination is relatively simple. A drop of fluid (of semen) is placed on a glass slide, then a drop of distilled water is added. A coverslip is placed over the preparation. The specimen is examined under a high-power microscope to determine the presence of sperm cells.
Other Characteristics of Semen
MALE AND FEMALE SPERM: There is an equal number of male and female sperm. The female sperm (X sperm) is slower, but stronger, while the male sperm (Y sperm) is faster but weaker.
SPERM’S LIFESPAN
It takes around 10 weeks for the sperm to reach maturity. Once the sperm is inside the woman’s body it can live up to 5 days or even longer under favorable conditions. However, when it is outside the body, it can only survive from a few minutes to several hours.
2. DRIED SPECIMEN
Examination most often required is done on the dry stain. Physical and chemical tests are used as preliminary tests. Confirmation is done by microscopic examination.
IMPORTANT NOTICE FOR THE INVESTIGATOR & FORENSIC EXAMINER: it is recommended that both blood and semen be “DRIED NATURALLY” to avoid destruction of the evidence.
3 METHODS OF Examination can be applied here:
[1] PHYSICAL EXAMINATION
A general visual examination for grayish-white or yellowish stain is first made. When dry, semen imparts a starchy stiffness to clothe. When inspected under ultraviolet light, seminal stain fluoresces. However, this fluorescence is not specific for semen only but may be observed from other materials or stains (LIKE BLOOD). The picture below is an example of detected HUMAN STAINS (can either be semen or other body fluids).

[2] CHEMICAL EXAMINATION
There are three chemical tests that can be used for seminal stains. They are the following:
(1) Florence Test (for detection of choline – seminal origin)
FLORENCE TEST is used in the confirmatory tests for seminal stain. The positive reaction reveals the presence of CHOLINE PERIODIDE CRYSTALS which are elongated and the color is brown. Florence test is discovered by DR. FLORENCE of Lyon, France.
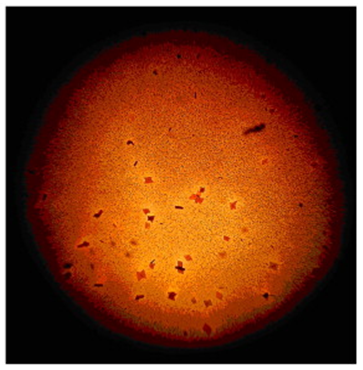
(2) Barberio’s Test (for detection of spermine – prostates origin)
BARBERIO’S test was discovered by Barberio in 1905. When the questioned stain is allowed to react with picric acid it leads to the formation of yellow needle-shaped spermine picrate crystals, which confirm the presence of seminal stains.
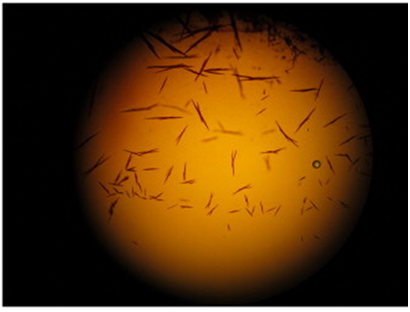
(3) Acid Phosphatase Test (for detection of acid phosphatase – prostate origin )
This is one of the BEST KNOWN and MOST WIDELY employed techniques for the purpose of identification of semen. This is very sensitive that even a person who had been vasectomized or azoospermic suspected of a sexual crime can be identified. The stability of the acid phosphatase in seminal stains depends on temperature. The drawback of this test is if the specimen is exposed to sunlight because the activity of acid phosphatase is reduced especially when exposed to heat over 60 degrees centigrade for 5 minutes, this test is NO LONGER EFFECTIVE.
When suspected stains are tested a color change to purple within the first 60 seconds is an INDICATIVE OF POSITIVE TEST.

The first two are based on the formation of characteristics crystals that are observed under the microscope. Acid phosphatase is an enzyme found in both animal and plant cells, but in large concentrations in human semen.
3. MICROSCOPIC EXAMINATION:
The only specific test for semen is the identification of sperm cells under a microscope. There are many factors, which may affect the detection of sperm cells, making this method difficult.
Some of these are the nature of the cloth in which the suspected stain is found, the age of the stain, the condition to which the stain was exposed, and the handling of the specimen.
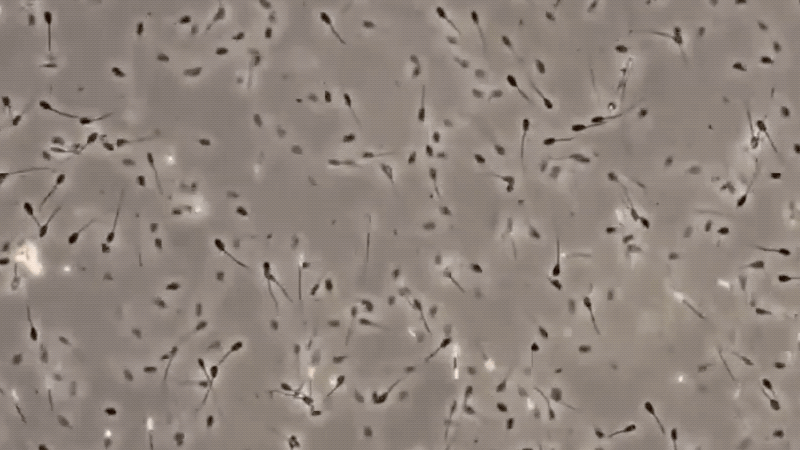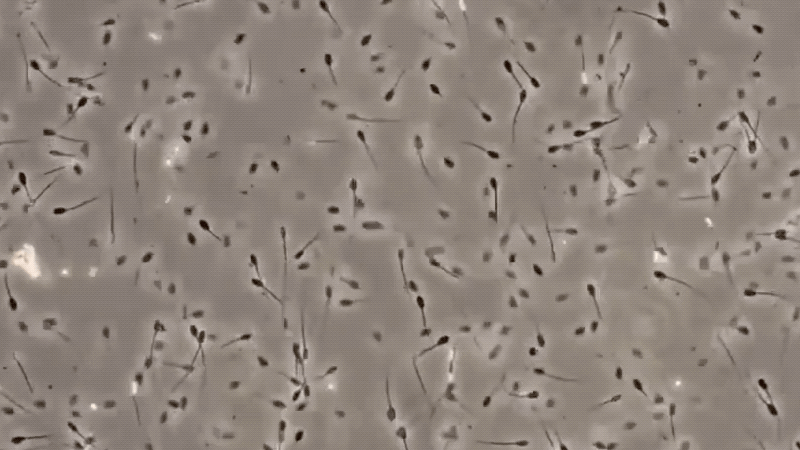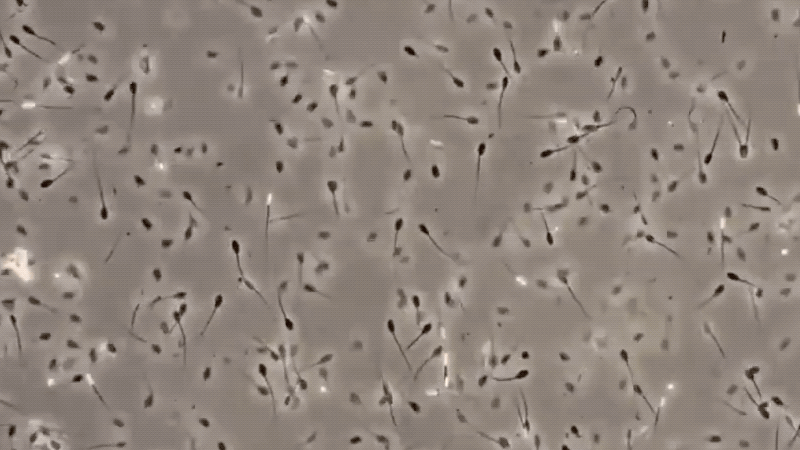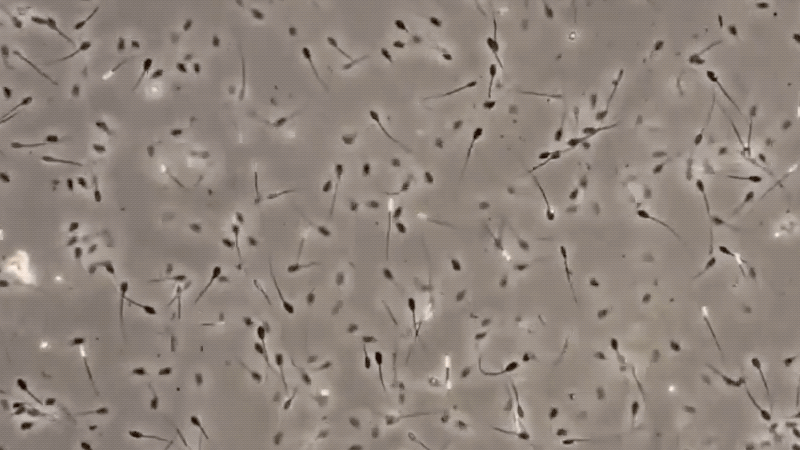
The presence of sperm cells proves that the stain is semen. However, with the absence of sperm cells, it cannot be concluded that the stain is not of seminal origin. These are some conditions that may be lead to non-detection of semen, like ASPERMIA, a disorder produced by the male organ which doesn’t produces or lack of semen, and OLIGOSPERMIA, semen with a very few count of sperm cells. Here are some of the definitive characteristics of semen and its defect:
Aspermia – complete lack of semen.
Asthenozoospermia – reduced sperm motility.
Azoospermia – an absence of sperm cells in semen.
Hyperspermia – large semen volume.
Hypospermia – small semen volume.
Oligospermia – few spermatozoa in semen.
Necrospermia – dead or immobile sperms.
Teratospermia – sperm with abnormal morphology.

We’ve ended our first module. GET READY FOR THE EXAMINATION! Just follow the arrow below.

This quiz is for logged in users only.
7 thoughts on “Forensic Chemistry Part 1”
Leave a Comment
You must be logged in to post a comment.

Robin Bautista
Yes
yes
Yes
Yes
Yes
Yes